MOLECULES OF TASTE - GINGEROL
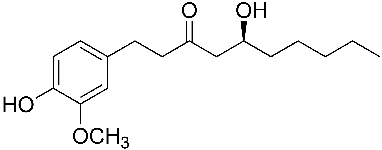
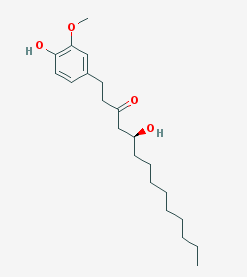
The 6-Gingerol  Molecule Molecule |
| 6-Gingerol | |
|---|---|
|
|
| Systematic name | (S)-5-hydroxy-1-(4-hydroxy-3-methoxyphenyl)-3-decanone |
| Molecular formula | C17H26O4 |
| SMILES | O=C(C[C@@H](O)CCCCC)CCc1cc(OC)c(O)cc1 |
| CAS number | [404-86-4] |
| Molecular mass | 294.38 g/mol |
| Melting point | 30 to 32 °C (86 to 90 °F; 303 to 305 K) |
Gingerol, or sometimes [6]-gingerol, is the active constituent of fresh ginger. Chemically, gingerol is a relative of capsaicin and piperine, the compounds which give chilli peppers and black pepper their respective spicyness. It is normally found as a pungent yellow oil, but also can form a low-melting crystalline solid.
Cooking ginger transforms gingerol into zingerone, which is less pungent and has a spicy-sweet aroma. When ginger is dried, gingerol undergoes a dehydration reaction forming shogaols, which are about twice as pungent as gingerol. This explains why dried ginger is more pungent than fresh ginger.
Ginger also contains 8-gingerol, 10-gingerol, and 12-gingerol.
Physiological effects
[6]-Gingerol administered by intraperitoneal injection has been used to induce a hypothermic state in rats.
Gingerol seems to be effective in an animal model of rheumatoid arthritis.
Gingerol has been investigated for its effect on cancerous tumors in the bowel, breast tissue, ovaries, the pancreas, among other tissues, with positive results.
The product is sometimes used for gingering of horses, a practice that is seen in the horse show world, and which is illegal in some, but not all disciplines.
|
The 10-Gingerol Molecule Fresh ginger extract exhibited a significant anti-neuroinflammatory capacity, which was largely owing to 10-gingerol, but not 6-gingerol. Anti-neuroinflammatory capacity of fresh ginger is attributed mainly to 10-gingerol. see abstract |
Tastant Molecules
Sugars
Artificial Sweeteners
Bitter Tastants
Umami
Acids
Thermal
Science of Chocolate

What are the health benefits of Chocolate?
What are the drugs in Chocolate?